The Basics
What are Ions? ions are atoms or molecules with a net electric charge, due to the loss or gain of one or more electrons!
How do they bond? And why do they care? atoms bond by sharing electrons until they maintain balance! Everyone wants to be balanced. That's when you find peace.
What three things am I required to know? You are required to know how to name ionic compounds, balance ionic compounds, and know how to write the Lewis Dot Structure of ionic compounds. I will explain how to do these below!
What you'll need.
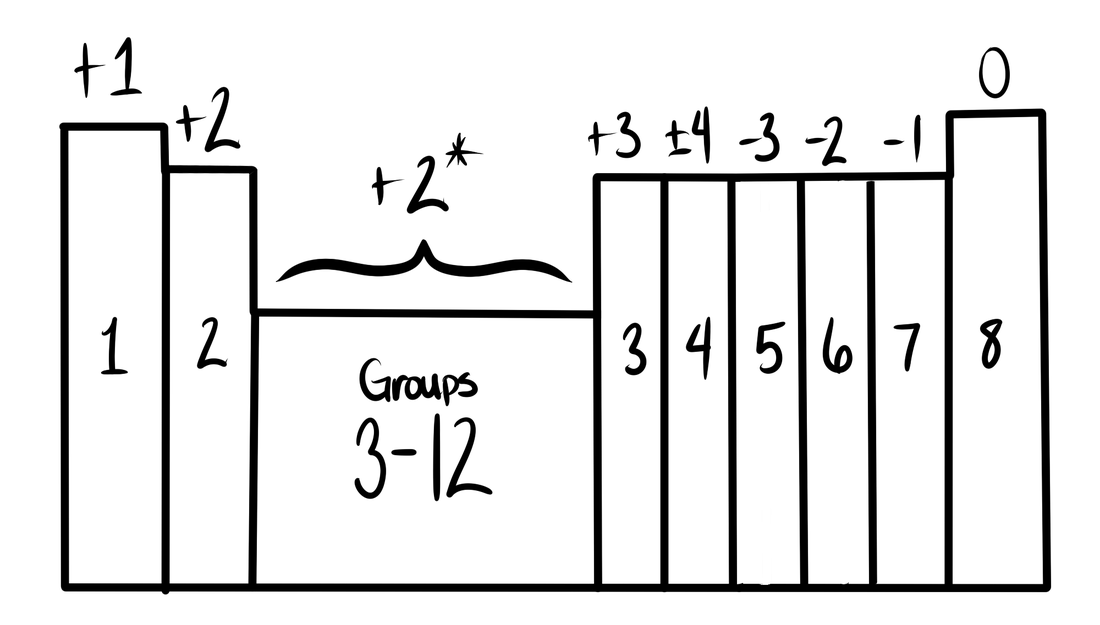
You will need a periodic table, and a table that shows the charges of each element.
Naming Ionic Compounds
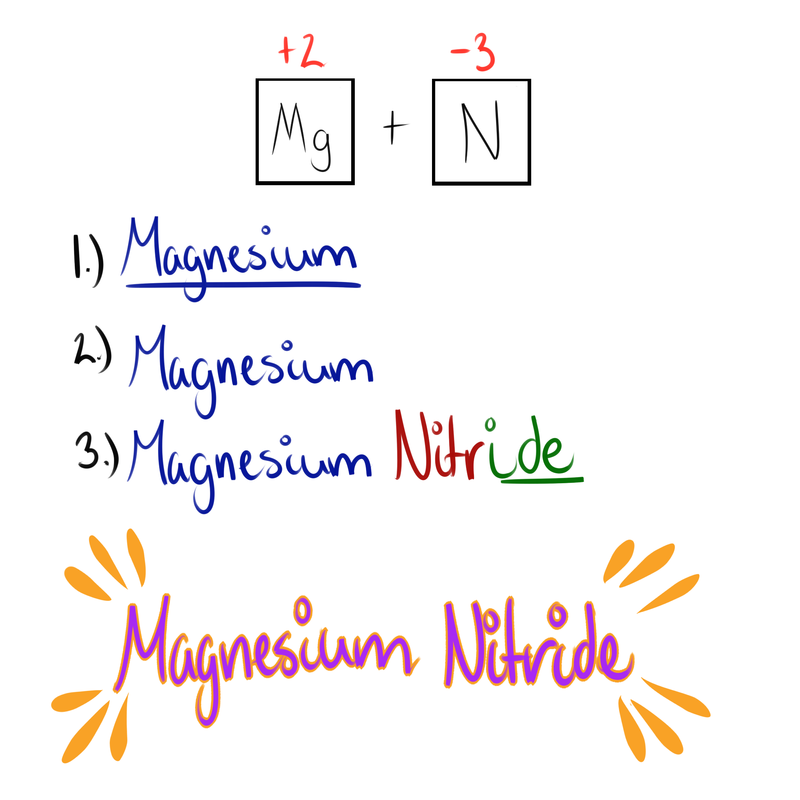
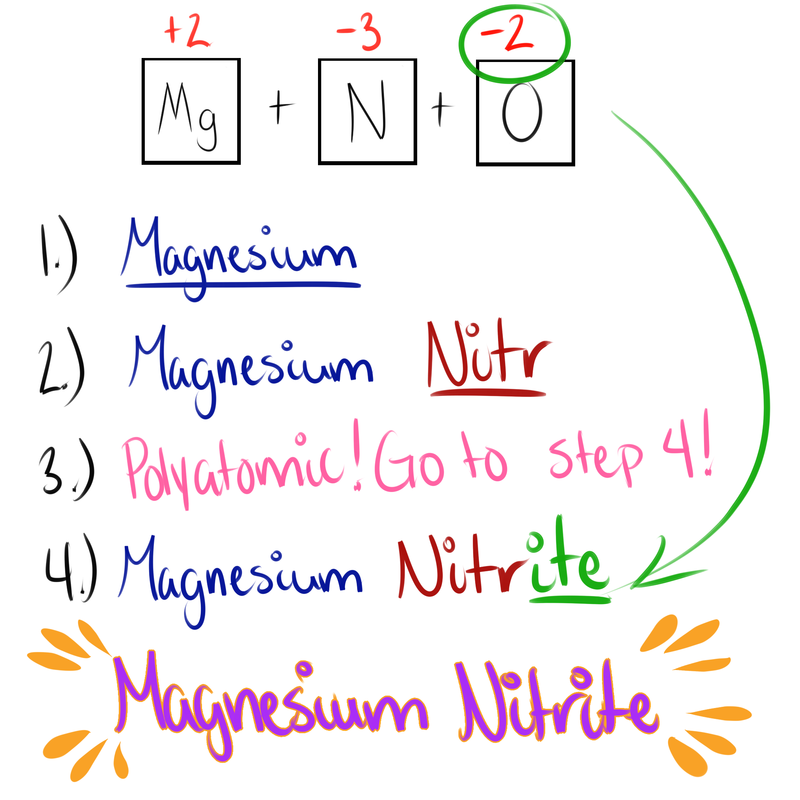
Here are the steps for naming Ionic Compounds!
- Write the name of the positive ion
- Write the root of the negative ion
- If binary (two elements) add the ending "-ide" if Polyatomic (more than two) then go to step 4
- If there is more oxygen present add "-ate" if there is less oxygen present add "-ite"
Balancing Ionic Compounds
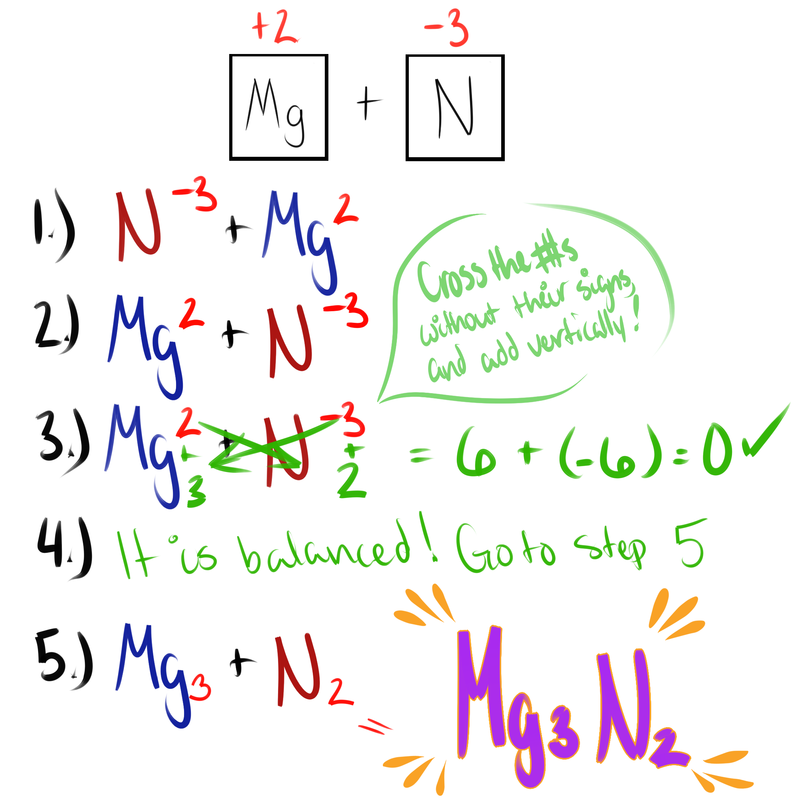
Here are the steps for balancing Ionic Compounds!
- Write the symbols of the ions with the oxidation numbers
- Rewrite with the positive ion first
- Check oxidation numbers for balance (if it's zero)
- If it is balanced (zero) go to step 5, if not, then oh no!
- Rewrite without the oxidation numbers.
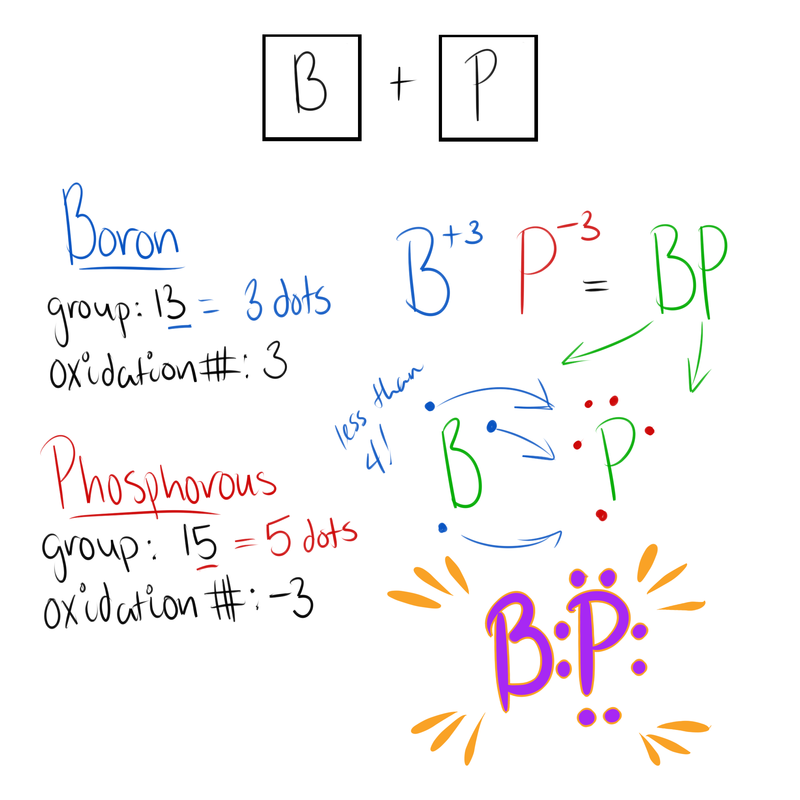
Writing the Lewis Dot Structure
Here are the steps for writing the Lewis Dot Structure of an Ionic Compound!
- Write down the elements and their group numbers
- The number of dots the element has = the group number
- Combine dots so that at least one of the elements has all 8 filled
- If an element has less than 4 dots, it will lose all of them! But if there are more than 4, then the element will be filled with all 8 dots
Extras
Special Case: Metallic Bonding
Metallic bonding is the electrostatic attraction between the positively charged nuclei of an atom in metal, and the de-localised electrons in metal. This often happens in an electron cloud or electron sea model. Basically, its the positive nucleus being attracted to stray electrons moving within the cloud of a metal.
Special Case: Electron Sea Model
The Electron Sea Model is a visual picture of delocalized electrons
Metallic bonding is the electrostatic attraction between the positively charged nuclei of an atom in metal, and the de-localised electrons in metal. This often happens in an electron cloud or electron sea model. Basically, its the positive nucleus being attracted to stray electrons moving within the cloud of a metal.
Special Case: Electron Sea Model
The Electron Sea Model is a visual picture of delocalized electrons
Work Cited
Definition of Ion: http://dictionary.reference.com/browse/ion?s=t
All pictures were made by ME
Metallic Bond: http://study.com/academy/lesson/what-is-a-metallic-bond-definition-properties-examples.html
I used my lab journal for instructions and other information.
All pictures were made by ME
Metallic Bond: http://study.com/academy/lesson/what-is-a-metallic-bond-definition-properties-examples.html
I used my lab journal for instructions and other information.